- Blog
- About
- Contact
- Serious Sam 1 Tpb
- Mocha Ae Cc Torrent
- Contoh Manual Prosedur Kerja Sekolah Menengah Kebangsaan
- Download Google Sketchup 8 Podium
- Timesupkidz Serial Number
- Addictive Drums Osx For Mac
- Mac Os X 10.7 Iso
- Driver Lan Dell Inspiron N4010 Win7 64bit
- Wii Iso Patcher Gui Version V1.2
- Hack Adsl Router
- Fire System Designer Bosch Software
- The Tiger And The Snow Torrent
- English 2015 Fast And Furious 7 Full Movie Utorrent
- Billion 7800nl Firmware Download
- Proxima Nova Light Italic Font
- Iso Buster Pro 2 4 0 1 Serial
- Sound Forge 7.0 Full Version Free Download
- Install Pdflib Linux
|
Writing a chemical (symbol) equation from a word equation? You'll have to identify the elements and compounds in the equation and snag the correct chemical symbols for each el ement and each compound. This might present a bit of a challenge, paticularly if there is a 'hydroxide' or a 'chlorate' or an 'oxalate' or something like that. In that case, you'll have to look up that anion or cation and see what elements make it up. Then you'll have to write up each reactant paying attention to insure you have the right proportions set down for your individual materials.
Shall we do a few examples? If you have oxygen you know that oxygen doesn't hang around by itself. I has a buddy, and is O2 in equations. Note that the subscript 2 is applied to tell us there are two atoms of oxygen in the diatomic molecule of this element. But, if you have sodium hydroxide, you'll know sodium in this compound is Na+ and the hydroxide isn't an element but and ion written as OH- in our expressions. It's oxygen and hydrogen (combined) with a -1 charge. Further, you'll have to account for the fact that Na and OH combine in a one-to-one ratio, and NaOH is sodium hydroxide.
In contrast, if you are given calcium hydroxide, Ca and OH combine in a one-to-two ratio, and Ca(OH)2 is the way we write this compound. It can get hairy, and you'll have to do some digging to insure you have the ions correctly written and the combination of elements set correctly in the expression of a compound. Once you've stepped through that, you can set up your equation. Certainly you'll have to know which materials are on the one side of the equation and which ones are on the other.
So now you've got the chemical expressions, and they're divided up into 'left side' and 'right side' groups. It's time to balance the equation. This may look daunting, but it's just a numbers game. It isn't totally dissimiliar from finding lowest common multiples for a group of numbers. Let's look at some examples. In the simple (classic!) acid-base reaction, an acid and a base react to yield a salt and water.
We'll use sodium hydroxide and hydrochloric acid mix them up to yield sodium chloride (table salt) and water. Crack office password recovery pro. Jujumao windows vista sp2 cn ultimate x64.2009.08. Sodium hydroxide is NaOH and hydrochloric acid is HCl. Let's hook them up and get the salt and water. Sodium hydroxide plus hydrochloric acid yields sodium chloride and water.
NaOH + HCl = NaCl + H2O We're not done. We've got our compounds correctly expressed throughout all the equation, and now we need to move on to balance the equation. We must insure everything on one side appears over on the second side. Each atom must be accounted for, every single one. So let's do that. There is one Na on each side. There is one O on each side.
There are 2 H's on each side, one each in the NaOH and the HCl, and 2 in the H2O on the other side. There is one Cl on each side, too. It's a balanced equation. We need to do one more. Here it is Calcium hydroxide plus sulfuric acid yields calcium sulfate plus water. Ca(OH)2 + H2SO4 = CaSO4 + H2O That's the correct expression of the materials, so let's move on to balance the equation. There is 1 Ca on each side.
There is an SO4 on each side, too. (We skipped to that ion, and treated it as a 'whole' unit.) The problem we have is that there are 4 hydrogens on the left side - 2 in the (OH)2 of the first compound, and 2 in H2 of the second compound - and only 2 of the H units on the right side, where they appear in the H2O there.
Cracking Equation
We need to balance this. We also have 2 atoms of O in the (OH)2 on the left, and only 1 of the O atoms on the right in the H2O there. (Note that we treated the SO4 as a unit, which we can do. In an acid-base reaction, we often 'shortcut' the process by doing that. We also treat water as H plus OH to make balancing easier, as the H is contributed by the acid and the OH is contributed by the base.) Moving on to balance the equation, all we have to do is double the number of water molecules to solve our problem.
That will raise the H and and the O atom count. Let's do that by writing a 2 in front of the water molecule and see how things look.
Ca(OH)2 + H2SO4 = CaSO4 + 2H2O Now we're cooking! There are 4 of the H atoms on the left and 4 on the right side. And there are now 2 of the O atoms on the left side (again, leaving the SO4 ion intact on both sides of the equation) and 2 of the O atoms in the water on the right side. Piece of cake. And you can do this. You can always balance an equation. Things may get 'fat' number wise, but it can always be balanced.
Just be sure you've reduced the numbers to lowest multiples so you don't end up with something out of control. The key to this is practice.
If you're rolling up your sleeves to begin chemistry, take this stuff to the bank. This is all there is to it, but you need to actually work a number of equations to get it down. And you'll also begin to learn the composition of the chemical ions (sometimes called radicals) like the hydroxide and the sulfate used as examples here. Easy as pie, and you can do this! Just remember to come back and answer a few questions here to pass on the knowledge. Good luck with your studies (but understand that luck is no substitute for study).
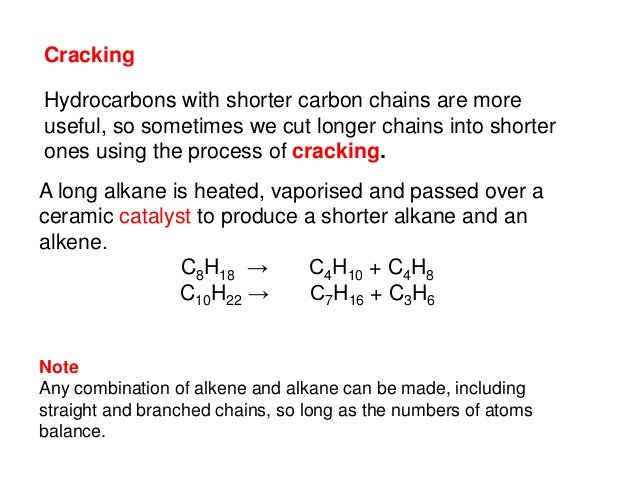
In order for it to be useful to us, crude oil is broken down in oil refineries into its component parts, known as fractions, and these can then be used for many different purposes. Fractions that are produced by the distillation distillation: Distillation is when we make a liquid evaporate and then condense the vapour back to a purer liquid.
Of crude oil can go through a process called cracking. This chemical reaction produces smaller hydrocarbons, including alkanes and alkenes.
Ethene and other alkenes are unsaturated unsaturated: An unsaturated compound contains at least one double or triple bond. hydrocarbons and can be used to make polymers. Ethene can be used to make ethanol. Cracking Fuels made from oil mixtures containing large hydrocarbon hydrocarbons: Hydrocarbons are a group of compounds which contain the elements hydrogen and carbon.
Molecules are not efficient. They do not flow easily and are difficult to ignite. Crude oil often contains too many large hydrocarbon molecules and not enough small hydrocarbon molecules to meet demand - this is where cracking comes in. Cracking allows large hydrocarbon molecules to be broken down into smaller, more useful hydrocarbon molecules. Fractions containing large hydrocarbon molecules are vaporised and passed over a hot catalyst catalyst: A catalyst changes the rate of a chemical reaction without being changed by the reaction itself.
This breaks chemical bonds in the molecules, and forms smaller hydrocarbon molecules. Cracking is an example of a thermal decomposition thermal decomposition: A reaction in which substances are broken down by heat. Kaspersky internet security 2011.
Comments are closed.
|
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
